Breast PET/CT
The development of a dedicated breast PET/CT scanner (in collaboration with the Cherry, Boone and Qi labs, the UC Davis Cancer Center and the Lawrence Berkeley National Lab)
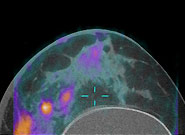
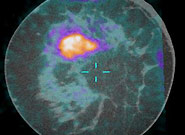
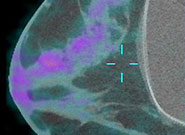
Breast cancer is a prevalent disease worldwide and is estimated to be the cause of more than 40,000 deaths in the United States in 2007 alone. Early diagnosis of breast cancer is extremely important, because treatment starting at a preliminary stage has been shown to significantly increase the 5-year patient survival rate.
Traditional mammography used for breast cancer imaging suffers from low sensitivity for women with dense breasts and tends to produce false-negative mammography results. We have designed a prototype PET component and have integrated it with a dedicated CT scanner constructed at our institution by Dr. John Boone’s research group.
This scanner allows for high resolution anatomical and metabolic imaging in breast cancer. Characterization studies using this scanner have been performed and currently, the scanner is undergoing clinical investigation. Preliminary results from the ongoing clinical trial are very promising.
High-resolution imaging in Rheumatoid Arthritis
The development of high-resolution molecular imaging techniques for Rheumatoid Arthritis (in collaboration with Cherry, Boone and Qi labs, the Imaging Research Center, the Departments of Internal Medicine and Radiation Oncology and the Clinical and Translational Sciences Center)

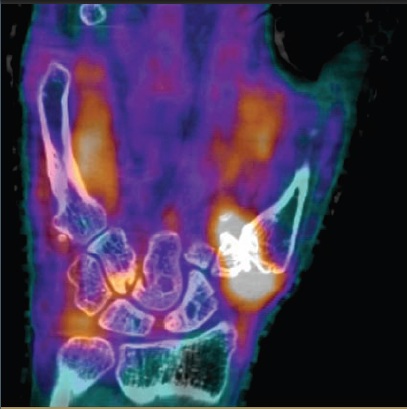
Rheumatoid arthritis is a debilitating inflammatory disease which results in 9 million physician visits and 250,000 hospitalizations per year. New molecularly-targeted pharmaceuticals are available to treat this disease, but clinical examination and standard imaging do not accurately distinguish long-term responders from non-responders to these expensive therapies. We are developing molecular imaging procedures that may separate responders from non-responders earlier than conventional methods using a unique high-resolution PET/CT scanner designed and built by UC Davis researchers and a high resolution MRI scanner. If early advanced imaging can predict outcomes, it will be possible to perform tests of therapeutic efficacy earlier in treatment, allowing those not responding to try alternative therapies prior to experiencing disease progression, joint damage, loss of function and debilitation. In addition, we anticipate that advanced imaging techniques such as those we are investigating may become accepted as surrogate end-points for clinical trials. This will also facilitate drug development for rheumatoid arthritis by reducing the time and costs needed to conduct such trials, with concomitant benefits to patients as their therapeutic options are widened.
Informatics tools for Tumor Metrics
The development of informatics tools to support image-based tumor metrics for assessing the response to cancer therapy (in collaboration with the Cancer Center and the Clinical and Translational Sciences Center)
We are developing an enterprise-level clinical trial support system for tumor metrics that includes the front end (data entry plug-in API interfaces with Philips iSite Enterprise PACS), the back end (Tumor Metrics database) and a web-based application that supports creating, retrieving and analyzing trial studies for clinical research coordinators. This interface is intended to support the following features: different levels of user accounts, data entry for trial creation and subject enrollment, and database queries for patients, trials, and lesion records. Future development includes tools for analysis of lesion change with plot diagrams and automatic generation of statistical reports.
Image segmentation tools for Tumor Metrics
The development and assessment of image segmentation tools for assessing tumor response (in collaboration with the Cancer Center and the Lawrence Livermore National Laboratory)
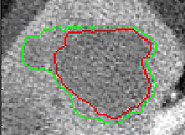
Measurement of lesion size change is an important indicator of response to therapy in cancer. However, current standard techniques (for example, RECIST) are simplistic, subject to operator error, labor-intensive and are not based on biological or imaging considerations. We are developing semi-automatic methods for lesion segmentation that may potentially perform at least as well as human observers in determining lesion volume in hepatic tumors. Our method is an iterative modification of the watershed method operating on a Gradient Vector Flow (GVF) transformation of the image. Our preliminary in vivo results show that our segmentation method has broadly similar accuracy to the physical observers. Our semi-automatic method is also expected to prove more time-efficient, repeatable and may prove to be a better predictor of outcome than manual measurements of tumor size change.
Large axial field-of-view PET scanners
The EXPLORER project (EXtreme Performance Long REsearch scanneR): Design explorations for a very large axial field of view PET scanner for humans (in collaboration with the Cherry and Qi labs at UC Davis, Bill Moses at Lawrence Berkeley National Lab and Joel Karp at the University of Pennsylvania)

Clinical whole-body PET scanners available today have an axial field of view (AFOV) ranging only from 15-25 cm, which limits sensitivity and prevents the acquisition of whole-body dynamic images. The objectives of this project are to determine the optimal design parameters and specifications for a 200 cm long PET scanner, and subsequently, to build one. The proposed scanner offers the possibility of whole-body dynamic PET imaging studies and will have vastly improved sesnitivity for cell tracking studies. It will also permit very low dose studies that can be used for longitudinal monitoring of pediatric and normal populations.
DOI encoding detectors for breast PET
The development of PSPMT/APD hybrid DOI detectors for breast PET (in collaboration with the Cherry Lab and RMD, inc.)

Breast PET scanners operate with the detectors very close to the object being imaged. This results in large parallax errors if the depth of photon interaction is not measured, with consequent loss of spatial resolution. We are developing hybrid DOI detectors using an approach related to that of Moses et al., in which scintillating crystal arrays are read out on the patient side by avalanche photodiodes and on the gantry side by position sensitive photomultiplier tubes. We have invested particular effort in reducing the dead space between the detectors, as system sensitivity is strongly adversely affected by this factor. Our current prototype devices use LSO, have a crystal pitch of 1.5 mm and a crystal depth of 20 mm. We hope to obtain depth of interaction resolution in the 2-3 mm range and energy resolution in the 15-17% range. The detectors we are designing should be well capable of operating in the high-flux environment encountered in breast imaging in humans.
Assessment of advanced 3D registration
3D registration for the spatial normalization of cross‐sectional and longitudinal high‐resolution breast PET/CT images
The performance of advanced 3D image registration (diffeomorphic demons) for normalizing cross-sectional pre- and post-contrast and longitudinal pre- and post contrast high resolution (HR) breast CT scans is being assessed. The former is for necessary for improving the delineation of breast tumors and accurately estimating the volume. The later is for monitoring neoadjuvant chemotherapy (NAC) in human breast cancer patients through the use of HR PET/CT.
Top row: Demons registration vs. rigid registration in longitudinal studies. Bottom row: Corresponding HR PET/CT scans where the PET image was transformed based on the warping field computed from the corresponding CT by the diffeomorphic demons method.

